The Publication:
Breast cancer cell-derived microRNA-155 suppresses tumor progression via enhancing immune cell recruitment and antitumor function
Sounds like a mouthful? Don’t worry—there’s a summary below.
Publication Summary:
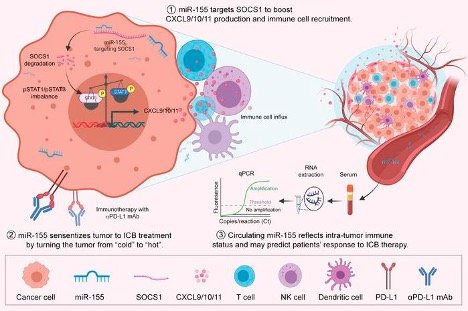
A small RNA, microRNA-155 (miR-155), has been shown to act against several types of cancer, including breast cancer. Previous evidence suggests that increased miR-155 expression in immune cells enhances host’s antitumor immune responses. However, given the reported association of miR-155 with tumorigenesis in various cancers, a debate is provoked on whether miR-155 is oncogenic or tumor suppressive. We aimed to interrogate the impact of tumor miR-155 expression, particularly that of cancer cell-derived miR-155, on antitumor immunity in breast cancer. We performed bioinformatic analysis of human breast cancer databases, murine experiments, and human specimen examination. We found that higher tumor miR-155 levels correlate with a favorable antitumor immune profile and better patient outcomes. Murine experiments demonstrated that miR-155 overexpression in breast cancer cells enhanced T cell influx, delayed tumor growth, and sensitized the tumors to immune checkpoint blockade (ICB) therapy. Mechanistically, miR-155 overexpression in breast cancer cells upregulated their CXCL9/10/11 production, which was mediated by SOCS1 inhibition and increased phosphorylated STAT1 (p-STAT1)/p-STAT3 ratios. We further found that serum miR-155 levels in breast cancer patients correlated with tumor miR-155 levels and tumor immune status. Our findings suggest that high serum and tumor miR-155 levels may be a favorable prognostic marker for breast cancer patients and that therapeutic elevation of miR-155 in breast tumors may improve the efficacy of immunotherapy via remodeling the antitumor immune landscape.